 It is primarily used in the production of semiconductors and LCD (Liquid Crystal Display) panels, as well as some types of solar panels and chemical lasers, but not all of them. It is used in the electronics industry and in high-power lasers because it is a source of fluorine. Nitrogen trifluoride is an odorless, colorless gas. The molar mass of Nitrogen is approximately 14.01 grams per mole of nitrogen atoms. It corresponds to the element’s atomic mass in AMU units (atomic mass units). The mass of an element in grams per mole of its atoms is known as its molar mass. To get a more detailed answer, click “how many cups in a gallon.” 8. In the United States, one cup equals half a pint (236.6 ml). How many cups in a gallon?Ī US liquid gallon is equal to 16 cups, and a US dry gallon is equal to 18.61 cups. Heavy water is used in nuclear power plants as a neutron moderator. Heavy water (D 2O), also known as deuterium oxide, is water made up of deuterium, a hydrogen isotope with twice the mass of ordinary hydrogen, and oxygen. 
Check full topic “How many electrons does oxygen have?”. Oxygen is a stable isotope of oxygen with a nucleus of 8 neutrons and 8 protons. H2S is polar or nonpolar?īecause of the presence of a lone pair of electrons in Sulfur and the electronegativity difference between Sulfur and H atoms, hydrogen sulfide is polar.Ī single oxygen atom has eight protons, eight electrons, and eight neutrons. Check the full article “How many hydrogen atoms are in a molecule of water?”. As a result, the covalent bonds are polar, and the oxygen atoms have a slight negative charge (due to the presence of an extra electron share), while the hydrogens have a slightly positive charge (from the extra un-neutralized protons). As a result, the electrons in a water molecule spend slightly more time around the oxygen atomic center and slightly less time around the hydrogen atomic center. Because oxygen atoms are electronegative, they attract the shared electrons in covalent bonds. Individual H2O molecules are V-shaped and are made up of two hydrogen atoms that are attached to the sides of a single oxygen atom. How many hydrogen atoms are in a molecule of water? In water, a hydrogen bond is a dynamic attraction between neighboring water molecules that involves one hydrogen atom located between two oxygen atoms. The hydrogen bond is a chemical bond formed by the hydrogen atom and more electronegative elements such as N, O, and F. Check the full article “How many neutrons does hydrogen have?”. Rare hydrogen isotopes, known as deuterium and tritium, have one and two neutrons, respectively. 
The majority of hydrogen atoms lack a neutron. Hydrogen is the most abundant element in the natureįrequently Asked Questions (FAQs) 1.Deuterium or heavy hydrogen contains one neutron.Natural hydrogen is a simple atom with one proton, one electron, and no neutron.H2 is a colorless, odorless, tasteless, non-toxic, and highly combustible gas.Hydrogen is a diatomic molecule with linear molecular geometry and 180-degree bond angles.īoth hydrogen atoms in the H2 molecule have identical electronegativity, and both atoms share equal ratios of bound shared electrons, resulting in a nonpolar molecule. Hydrogen gas is a loose aggregation of hydrogen molecules, each of which has two atoms, making it a diatomic molecule, H2. The nucleus of a hydrogen atom is made up of one proton with one unit of positive electrical charge and one electron with one unit of negative electrical charge. 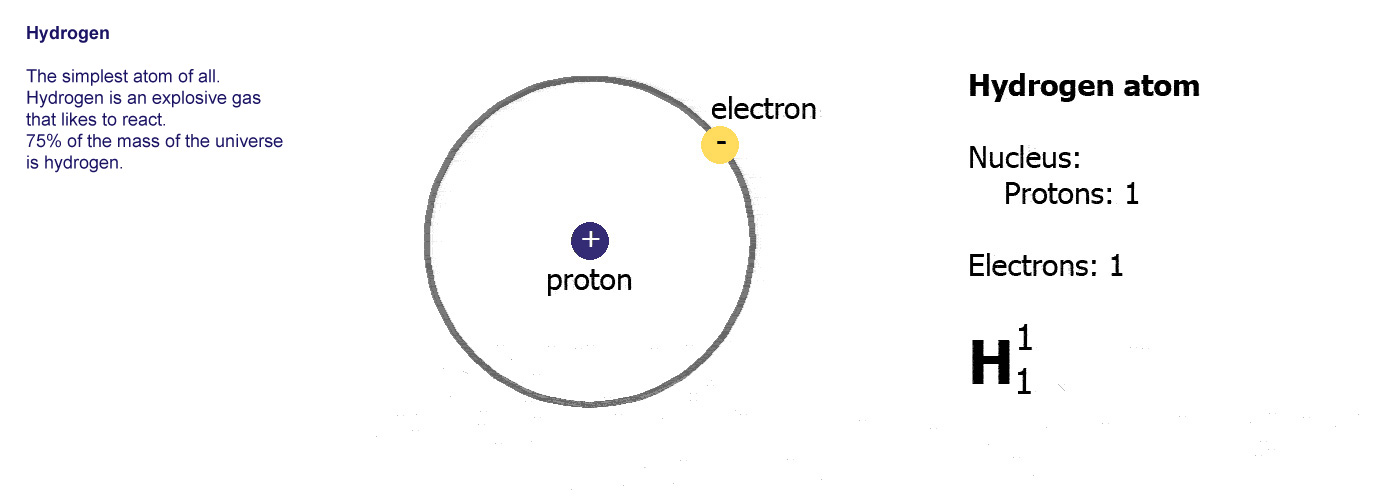
Hydrogen (H) is the most fundamental member of the chemical element family and is a colorless, odorless, tasteless, flammable gaseous substance. Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75 percent of all normal matter. 
It is colorless, odorless, tasteless, non-toxic, and highly combustible. At standard conditions, hydrogen is a gas of diatomic molecules having the formula H 2. Hydrogen is the chemical element with the symbol H and atomic number 1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
